What are Cosmic Rays?

Cosmic rays are numerous types of ionizing radiation being emitted from different sources, all in outer space. If it were not for the protective magnetosphere and atmosphere protecting Earth’s surface, life would have had a difficult time surviving or being complex or diverse.
The term cosmic rays does not specifically refer to any type electromagnetic radiation comprised of photons such as gamma rays, radio waves, or light (which are also abundant in space).
Types of Cosmic Rays
Galactic Cosmic Rays (from within the Milky Way) and Extragalactic Cosmic Rays (from outside of our galaxy) produced by celestial bodies and/or events such as supernovae in deep space.
Solar Cosmic Rays (also called Solar Energetic Particles) emitted from our own sun, Sol, primarily during solar flares or coronal mass ejections.
Context determines the likelihood of which type of radiation to which people are referring when they use the term Solar Rays.
Composition
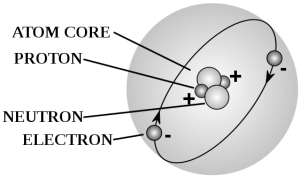
Like alpha radiation we often encounter on Earth from unstable elements undergoing nuclear decay, about 99% of cosmic rays are comprised of relatively large atomic particles structurally equivalent to the nucleus of an atom (an atom without its electron shell). Alpha rays are made of particles the same as a helium nucleus, and around 9% of these typical cosmic rays are the same. Around 90% are simple protons equivalent to the nucleus of a hydrogen atom, and about 1% are nuclei of heavier (larger) atoms.
There is about 1% of cosmic rays which are beta radiation (an electron) and/or antimatter (positrons or anti-protons).
Effects of Cosmic Radiation
Astronauts are exposed to relatively high amounts of cosmic rays and it has even been theorized that cherry seeds brought back from 9 months in the International Space Station sprouted, grew, and bloomed differently than seeds from the parent tree should. One reason there are no control seeds to which to compare them is that no other seeds from the 1000+ year-old tree had ever even sprouted, whereas 4 of the “space seeds” did and bloomed years ahead of schedule. The endeavor was initially just to excite people about seeds that had been in space; it wasn’t really wasn’t intended much of an actual experiment (esp. since much more controlled experiments such as with potatoes were already done, yielding few interesting results). A controlled experiment will have to be performed to draw more solid hypotheses of whether or not 8 months of microgravity and cosmic radiation truly affected the cherry seeds or not.
Cosmic rays can interfere with electronics and pose an increased risk of cancer. Even commercial airline crew are exposed to extra solar radiation, being frequently above much of Earths atmosphere which helps to shield the radiation.
What are Cosmic Rays? Read More »